What Information Is Needed to Determine the General Shape of an Orbital
2.2: Diminutive Orbitals and Quantum Numbers
- Page ID
- 122444
Skills to Develop
- Sympathize the general thought of the quantum mechanical description of electrons in an cantlet, and that information technology uses the notion of three-dimensional wave functions, or orbitals, that define the distribution of probability to observe an electron in a particular office of space
- List and describe traits of the 4 quantum numbers that grade the ground for completely specifying the country of an electron in an cantlet
Understanding Quantum Theory of Electrons in Atoms
Video \(\PageIndex{1}\) : A preview of electrons in orbitals.
The goal of this department is to understand the electron orbitals (location of electrons in atoms), their dissimilar energies, and other properties. The use of quantum theory provides the best agreement to these topics. This cognition is a precursor to chemic bonding.
Every bit was described previously, electrons in atoms can exist only on discrete energy levels but not between them. Information technology is said that the energy of an electron in an atom is quantized, that is, it tin exist equal just to certain specific values and can jump from 1 free energy level to another but non transition smoothly or stay betwixt these levels.
The energy levels are labeled with an northward value, where northward = 1, ii, 3, …. Generally speaking, the energy of an electron in an atom is greater for greater values of n. This number, n, is referred to equally the principal quantum number. The main quantum number defines the location of the energy level. It is essentially the same concept as the n in the Bohr atom description. Another name for the principal quantum number is the shell number. The shells of an cantlet can be thought of concentric circles radiating out from the nucleus. The electrons that belong to a specific shell are most likely to be establish inside the respective circular area. The further we proceed from the nucleus, the higher the shell number, and and so the higher the energy level (Figure \(\PageIndex{i}\)). The positively charged protons in the nucleus stabilize the electronic orbitals by electrostatic attraction between the positive charges of the protons and the negative charges of the electrons. So the farther away the electron is from the nucleus, the greater the energy information technology has.
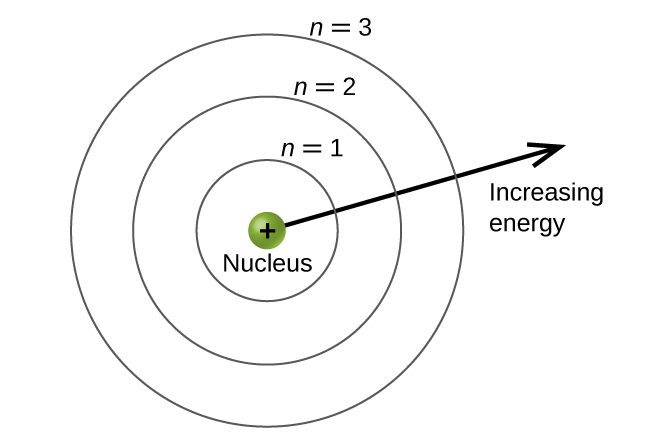
Figure \(\PageIndex{1}\) : Different shells are numbered by chief quantum numbers.
This breakthrough mechanical model for where electrons reside in an atom can be used to await at electronic transitions, the events when an electron moves from one energy level to another. If the transition is to a higher energy level, energy is absorbed, and the energy alter has a positive value. To obtain the amount of free energy necessary for the transition to a college energy level, a photon is captivated past the atom. A transition to a lower energy level involves a release of energy, and the free energy change is negative. This process is accompanied by emission of a photon by the atom. The following equation summarizes these relationships and is based on the hydrogen atom:
\[ \begin{align*} ΔE &=E_\ce{concluding}−E_\ce{initial} \\[5pt] &=−2.18×x^{−18}\left(\dfrac{1}{due north^2_\ce f}−\dfrac{1}{due north^2_\ce i}\correct)\:\ce J \end{align*} \]
The values north f and n i are the final and initial free energy states of the electron.
The primary quantum number is 1 of three quantum numbers used to characterize an orbital. An atomic orbital, which is distinct from an orbit, is a general region in an cantlet within which an electron is most probable to reside. The quantum mechanical model specifies the probability of finding an electron in the three-dimensional infinite around the nucleus and is based on solutions of the Schrödinger equation. In addition, the principal quantum number defines the energy of an electron in a hydrogen or hydrogen-similar atom or an ion (an atom or an ion with only ane electron) and the general region in which discrete energy levels of electrons in a multi-electron atoms and ions are located.
Another quantum number is l, the athwart momentum quantum number. It is an integer that defines the shape of the orbital, and takes on the values, l = 0, one, two, …, n – ane. This ways that an orbital with n = ane can accept just one value of l, l = 0, whereas n = 2 permits l = 0 and l = i, and so on. The chief quantum number defines the full general size and energy of the orbital. The fifty value specifies the shape of the orbital. Orbitals with the same value of l course a subshell. In addition, the greater the angular momentum breakthrough number, the greater is the angular momentum of an electron at this orbital.
Orbitals with fifty = 0 are called south orbitals (or the s subshells). The value l = one corresponds to the p orbitals. For a given n, p orbitals constitute a p subshell (eastward.thousand., 3p if n = 3). The orbitals with l = 2 are called the d orbitals, followed by the f-, thousand-, and h-orbitals for l = 3, four, five, and at that place are higher values we volition non consider.
There are sure distances from the nucleus at which the probability density of finding an electron located at a particular orbital is aught. In other words, the value of the wavefunction ψ is zilch at this distance for this orbital. Such a value of radius r is called a radial node. The number of radial nodes in an orbital is north – 50 – 1.
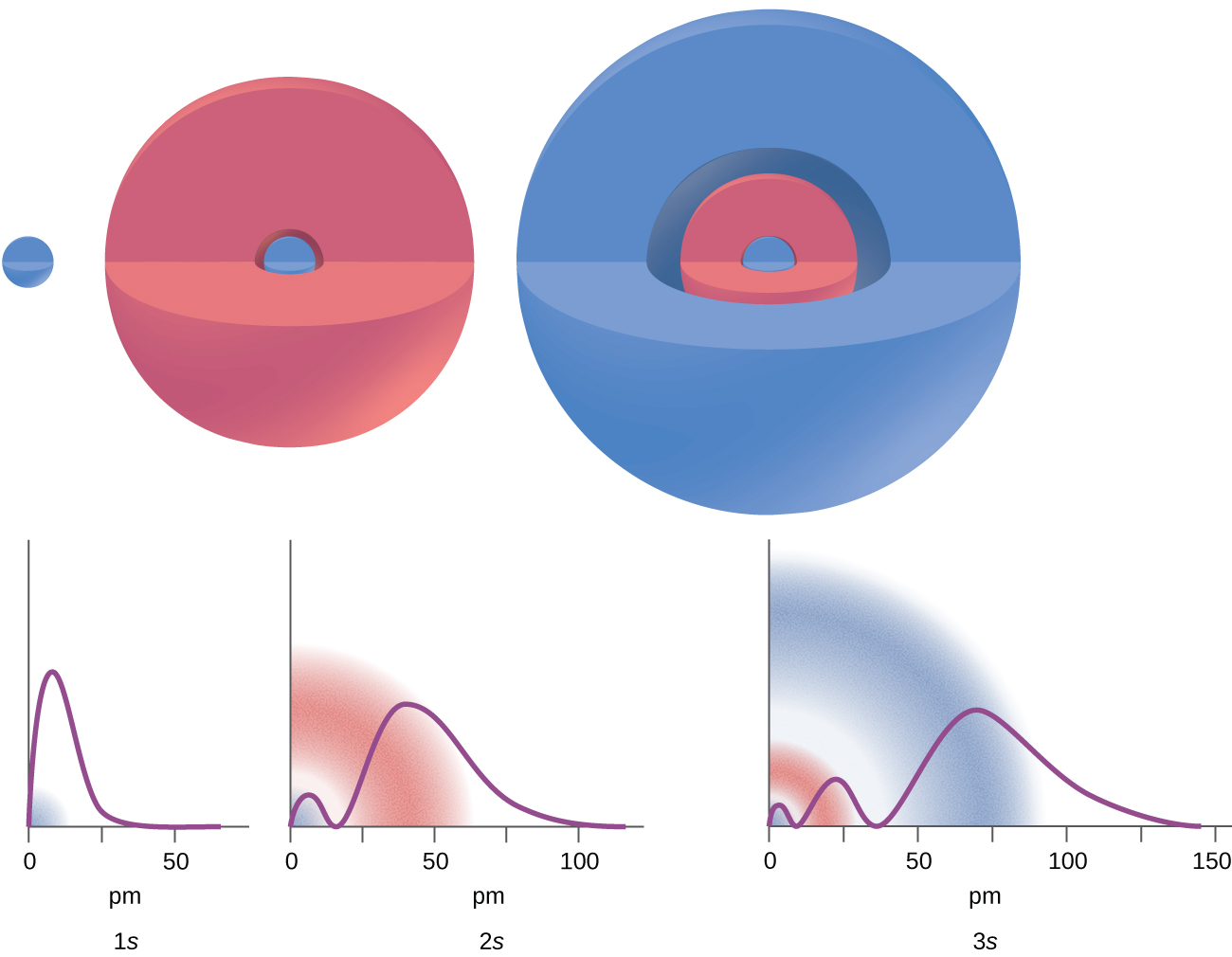
Effigy \(\PageIndex{2}\) : The graphs evidence the probability (y axis) of finding an electron for the 1s, 2s, 3s orbitals every bit a function of distance from the nucleus.
Video \(\PageIndex{2}\) : Looking into the probability of finding electrons.
Consider the examples in Figure \(\PageIndex{3}\). The orbitals depicted are of the s blazon, thus l = 0 for all of them. Information technology can be seen from the graphs of the probability densities that there are 1 – 0 – i = 0 places where the density is naught (nodes) for 1s (n = 1), ii – 0 – 1 = 1 node for 2southward, and 3 – 0 – one = two nodes for the 3southward orbitals.
The s subshell electron density distribution is spherical and the p subshell has a dumbbell shape. The d and f orbitals are more complex. These shapes stand for the 3-dimensional regions within which the electron is probable to exist found.
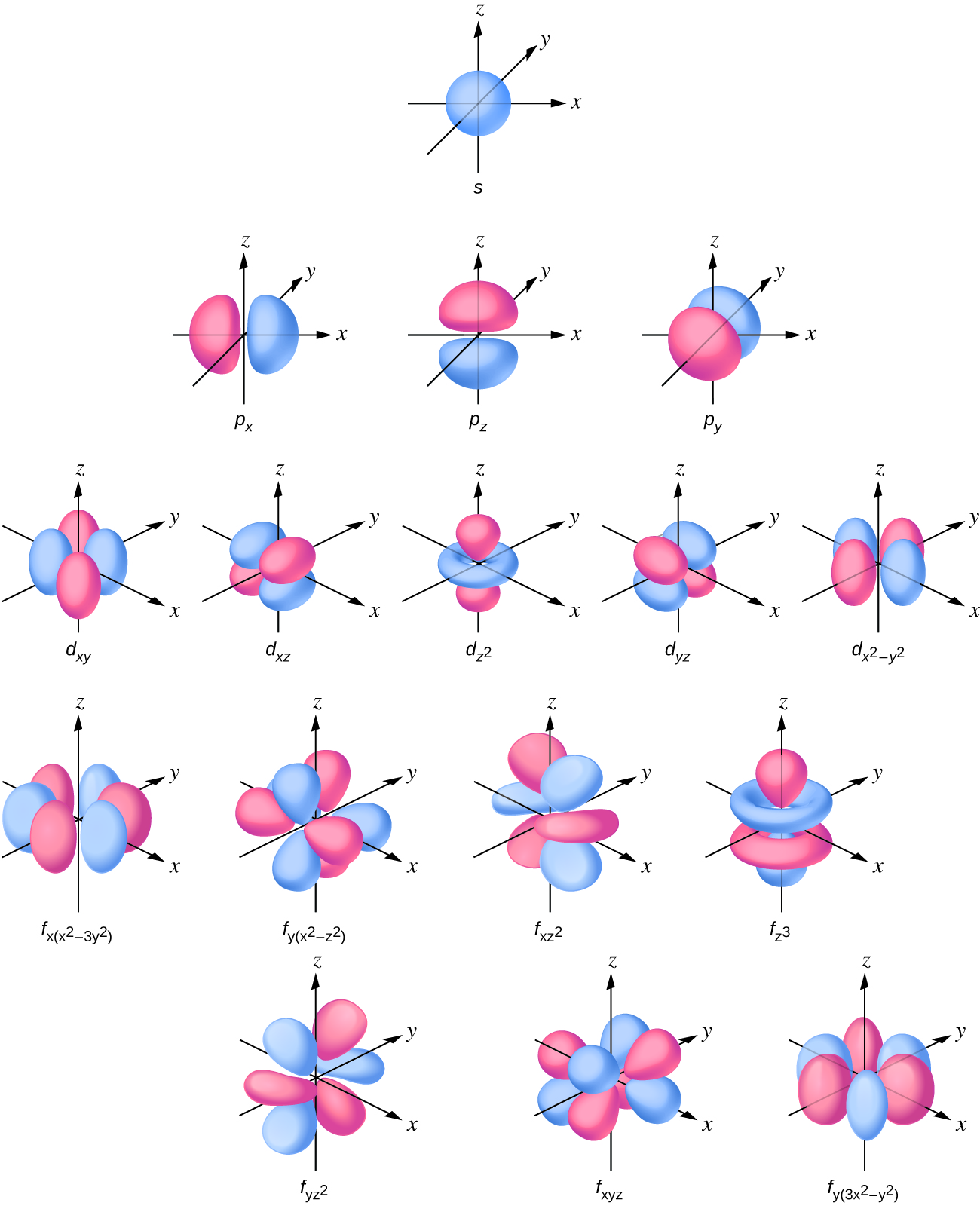
Figure \(\PageIndex{3}\) : Shapes of due south, p, d, and f orbitals.
If an electron has an angular momentum (l ≠ 0), and then this vector can indicate in dissimilar directions. In addition, the z component of the angular momentum can have more than one value. This means that if a magnetic field is applied in the z direction, orbitals with different values of the z component of the athwart momentum volition accept different energies resulting from interacting with the field. The magnetic quantum number, called m50, specifies the z component of the angular momentum for a detail orbital. For example, for an due south orbital, l = 0, and the only value of gfifty is zero. For p orbitals, 50 = one, and ml can be equal to –i, 0, or +1. Mostly speaking, chiliadfifty tin can be equal to –50, –(l – 1), …, –1, 0, +1, …, (50 – i), l. The total number of possible orbitals with the aforementioned value of fifty (a subshell) is 2l + 1. Thus, there is one due south-orbital for ml = 0, there are three p-orbitals for ml = 1, five d-orbitals for ml = ii, vii f-orbitals for ml = 3, and so forth. The principal quantum number defines the general value of the electronic free energy. The angular momentum quantum number determines the shape of the orbital. And the magnetic breakthrough number specifies orientation of the orbital in space, equally tin exist seen in Figure \(\PageIndex{iii}\).
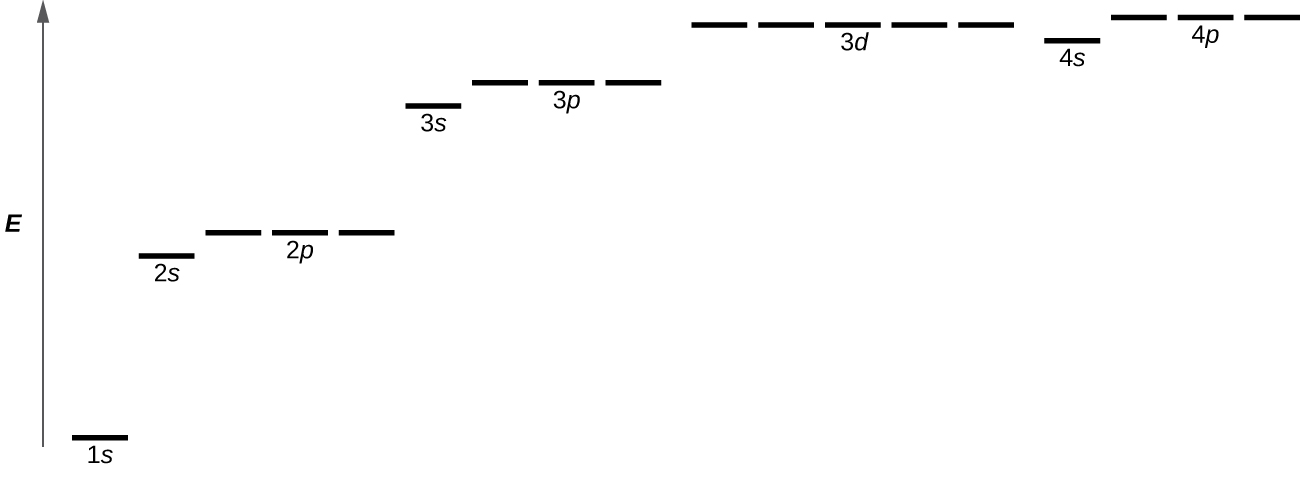
Figure \(\PageIndex{4}\) : The chart shows the energies of electron orbitals in a multi-electron atom.
Figure \(\PageIndex{4}\) illustrates the free energy levels for various orbitals. The number earlier the orbital name (such as 2due south, 3p, and then forth) stands for the principal quantum number, northward. The letter in the orbital name defines the subshell with a specific athwart momentum breakthrough number l = 0 for s orbitals, 1 for p orbitals, 2 for d orbitals. Finally, there are more than one possible orbitals for l ≥ i, each corresponding to a specific value of thoufifty . In the instance of a hydrogen atom or a one-electron ion (such every bit He+, Li2 +, and so on), energies of all the orbitals with the same due north are the same. This is called a degeneracy, and the energy levels for the same principal breakthrough number, n, are chosen degenerate energy levels. Yet, in atoms with more than than one electron, this degeneracy is eliminated by the electron–electron interactions, and orbitals that belong to different subshells have different energies. Orbitals within the same subshell (for example ns, np, nd, nf, such as 2p, 3s) are still degenerate and have the same energy.
While the three quantum numbers discussed in the previous paragraphs work well for describing electron orbitals, some experiments showed that they were not sufficient to explain all observed results. It was demonstrated in the 1920s that when hydrogen-line spectra are examined at extremely high resolution, some lines are actually not single peaks merely, rather, pairs of closely spaced lines. This is the so-called fine structure of the spectrum, and information technology implies that there are boosted small-scale differences in energies of electrons fifty-fifty when they are located in the aforementioned orbital. These observations led Samuel Goudsmit and George Uhlenbeck to propose that electrons take a quaternary quantum number. They chosen this the spin quantum number, or mdue south .
The other three quantum numbers, north, l, and ml , are properties of specific atomic orbitals that too define in what role of the infinite an electron is about likely to be located. Orbitals are a result of solving the Schrödinger equation for electrons in atoms. The electron spin is a dissimilar kind of holding. It is a completely quantum phenomenon with no analogues in the classical realm. In addition, it cannot be derived from solving the Schrödinger equation and is not related to the normal spatial coordinates (such as the Cartesian x, y, and z). Electron spin describes an intrinsic electron "rotation" or "spinning." Each electron acts every bit a tiny magnet or a tiny rotating object with an athwart momentum, fifty-fifty though this rotation cannot be observed in terms of the spatial coordinates.
The magnitude of the overall electron spin can just have one value, and an electron tin only "spin" in one of 2 quantized states. One is termed the α land, with the z component of the spin being in the positive direction of the z axis. This corresponds to the spin breakthrough number
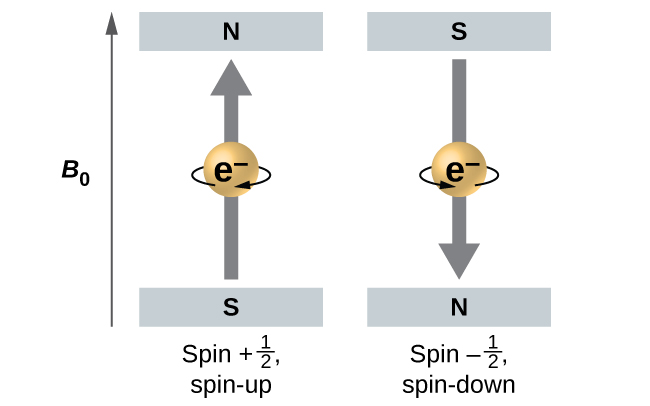
Effigy \(\PageIndex{5}\) : Electrons with spin values \(
Figure \(\PageIndex{v}\) illustrates this phenomenon. An electron acts like a tiny magnet. Its moment is directed upwards (in the positive direction of the z centrality) for the
Video \(\PageIndex{three}\) : The incertitude of the location of electrons.
The Pauli Exclusion Principle
An electron in an cantlet is completely described by four quantum numbers: n, 50, yardl , and yardsouthward . The first three quantum numbers define the orbital and the quaternary breakthrough number describes the intrinsic electron belongings called spin. An Austrian physicist Wolfgang Pauli formulated a full general principle that gives the last piece of data that we need to understand the general behavior of electrons in atoms. The Pauli exclusion principle tin exist formulated as follows: No two electrons in the same atom tin have exactly the same set of all the four breakthrough numbers. What this means is that electrons can share the same orbital (the same set of the quantum numbers due north, l, and mfifty ), but merely if their spin breakthrough numbers ks have unlike values. Since the spin quantum number tin only have two values
| Proper noun | Symbol | Allowed values | Physical pregnant |
|---|---|---|---|
| principal quantum number | n | 1, 2, 3, 4, …. | shell, the general region for the value of energy for an electron on the orbital |
| angular momentum or azimuthal quantum number | 50 | 0 ≤ 50 ≤ n – one | subshell, the shape of the orbital |
| magnetic quantum number | ml | – fifty ≤ ml ≤ fifty | orientation of the orbital |
| spin breakthrough number | ks | \(\dfrac{1}{2},\:−\dfrac{1}{2}\) | direction of the intrinsic quantum "spinning" of the electron |
Example \(\PageIndex{i}\): Working with Shells and Subshells
Point the number of subshells, the number of orbitals in each subshell, and the values of l and ml for the orbitals in the n = 4 trounce of an cantlet.
Solution
For n = iv, l tin can have values of 0, 1, 2, and iii. Thus, s, p, d, and f subshells are constitute in the north = iv shell of an atom. For l = 0 (the southward subshell), m50 can only exist 0. Thus, there is only i 4s orbital. For fifty = i (p-blazon orbitals), one thousand tin take values of –i, 0, +1, and so we notice 3 4p orbitals. For l = 2 (d-type orbitals), ml can have values of –2, –1, 0, +1, +2, so we take 5 4d orbitals. When 50 = 3 (f-type orbitals), gl can have values of –3, –ii, –1, 0, +1, +2, +3, and we can have vii 4f orbitals. Thus, we find a total of sixteen orbitals in the north = four beat of an atom.
Exercise \(\PageIndex{one}\)
Identify the subshell in which electrons with the following breakthrough numbers are constitute:
- north = 3, l = i;
- north = v, l = three;
- n = ii, l = 0.
- Respond a
-
3p
- Answer b
-
5f
- Answer c
-
2s
Example \(\PageIndex{2}\): Maximum Number of Electrons
Calculate the maximum number of electrons that can occupy a shell with (a) northward = ii, (b) due north = five, and (c) n as a variable. Note you are only looking at the orbitals with the specified north value, not those at lower energies.
Solution
(a) When due north = ii, there are 4 orbitals (a unmarried twos orbital, and three orbitals labeled twop). These four orbitals can contain eight electrons.
(b) When n = 5, at that place are five subshells of orbitals that we need to sum:
\(\begin{align*}
&\phantom{+}\textrm{1 orbital labeled }5s\\
&\phantom{+}\textrm{3 orbitals labeled }5p\\
&\phantom{+}\textrm{5 orbitals labeled }5d\\
&\phantom{+}\textrm{vii orbitals labeled }5f\\
&\underline{+\textrm{nine orbitals labeled }5g}\\
&\,\textrm{25 orbitals total}
\end{align*}\)
Once more, each orbital holds ii electrons, so 50 electrons tin can fit in this trounce.
(c) The number of orbitals in whatsoever shell north will equal n 2 . There can be up to two electrons in each orbital, so the maximum number of electrons will be 2
Practise \(\PageIndex{2}\)
If a shell contains a maximum of 32 electrons, what is the main quantum number, n?
- Respond
-
n = 4
Example \(\PageIndex{3}\): Working with Breakthrough Numbers
Complete the following table for atomic orbitals:
| Orbital | northward | fifty | kl degeneracy | Radial nodes (no.) |
|---|---|---|---|---|
| 4f | ||||
| iv | 1 | |||
| 7 | 7 | 3 | ||
| vd |
Solution
The tabular array can exist completed using the following rules:
- The orbital designation is nl, where fifty = 0, 1, 2, 3, 4, 5, … is mapped to the letter sequence s, p, d, f, g, h, …,
- The yardfifty degeneracy is the number of orbitals within an l subshell, and and so is 2l + ane (there is ane s orbital, three p orbitals, five d orbitals, 7 f orbitals, so along).
- The number of radial nodes is equal to n – fifty – ane.
| Orbital | n | l | chiliadl degeneracy | Radial nodes (no.) |
|---|---|---|---|---|
| ivf | iv | 3 | seven | 0 |
| 4p | 4 | ane | 3 | 2 |
| 7f | vii | iii | seven | iii |
| 5d | 5 | two | 5 | 2 |
Exercise \(\PageIndex{3}\)
How many orbitals take l = 2 and n = 3?
- Answer
-
The five degenerate 3d orbitals
Summary
Video \(\PageIndex{4}\) : An overview of orbitals.
An diminutive orbital is characterized past three quantum numbers. The principal breakthrough number, n, can be any positive integer. The general region for value of energy of the orbital and the average distance of an electron from the nucleus are related to north. Orbitals having the aforementioned value of due north are said to be in the same shell. The angular momentum quantum number, l, can have any integer value from 0 to n – 1. This quantum number describes the shape or type of the orbital. Orbitals with the aforementioned principal breakthrough number and the same l value vest to the same subshell. The magnetic breakthrough number, gfifty , with 2fifty + 1 values ranging from –l to +fifty, describes the orientation of the orbital in infinite. In improver, each electron has a spin quantum number, gsouthward , that can exist equal to \(±\dfrac{1}{2}\) . No two electrons in the same cantlet tin have the same set of values for all the four quantum numbers.
Glossary
- angular momentum quantum number (l)
- quantum number distinguishing the different shapes of orbitals; it is likewise a measure of the orbital angular momentum
- wavefunction (ψ)
- mathematical clarification of an diminutive orbital that describes the shape of the orbital; it can exist used to summate the probability of finding the electron at whatever given location in the orbital, equally well as dynamical variables such equally the energy and the angular momentum
- subshell
- set of orbitals in an atom with the same values of n and l
- spin breakthrough number (thousands )
- number specifying the electron spin management, either \(+\dfrac{ane}{two}\)
or \(−\dfrac{i}{ii}\) - beat out
- set up of orbitals with the same principal breakthrough number, north
- due south orbital
- spherical region of space with high electron density, describes orbitals with fifty = 0. An electron in this orbital is called an s electron
- quantum mechanics
- discipline that includes quantization of energy, wave-particle duality, and the Heisenberg incertitude principle to draw matter
- principal quantum number (n)
- breakthrough number specifying the shell an electron occupies in an atom
- Pauli exclusion principle
- specifies that no two electrons in an atom tin can have the aforementioned value for all four breakthrough numbers
- p orbital
- dumbbell-shaped region of infinite with high electron density, describes orbitals with fifty = one. An electron in this orbital is called a p electron
- magnetic quantum number (ml )
- breakthrough number signifying the orientation of an atomic orbital effectually the nucleus; orbitals having different values of ml but the same subshell value of l take the same energy (are degenerate), but this degeneracy can be removed by awarding of an external magnetic field
- Heisenberg dubiety principle
- rule stating that it is impossible to exactly determine both certain cohabit dynamical properties such as the momentum and the position of a particle at the same time. The uncertainty principle is a issue of quantum particles exhibiting moving ridge–particle duality
- f orbital
- multilobed region of space with high electron density, describes orbitals with fifty = three. An electron in this orbital is chosen an f electron
- electron density
- a measure out of the probability of locating an electron in a particular region of space, it is equal to the squared absolute value of the wave function ψ
- d orbital
- region of space with high electron density that is either four lobed or contains a dumbbell and torus shape; describes orbitals with 50 = two. An electron in this orbital is called a d electron
- atomic orbital
- mathematical office that describes the behavior of an electron in an atom (also chosen the wavefunction), it can be used to observe the probability of locating an electron in a specific region around the nucleus, as well as other dynamical variables
Contributors
-
Paul Flowers (Academy of North Carolina - Pembroke), Klaus Theopold (University of Delaware) and Richard Langley (Stephen F. Austin State Academy) with contributing authors.Textbook content produced by OpenStax Higher is licensed under a Creative Commons Attribution License 4.0 license. Download for free at http://cnx.org/contents/85abf193-2bd...a7ac8df6@9.110).
- Adelaide Clark, Oregon Institute of Technology
- Crash Course Physics: Crash Course is a division of Complexly and videos are gratis to stream for educational purposes.
- Crash Course Chemistry: Crash Class is a segmentation of Complexly and videos are free to stream for educational purposes.
- TED-Ed's commitment to creating lessons worth sharing is an extension of TED's mission of spreading great ideas. Inside TED-Ed'south growing library of TED-Ed animations, y'all will find carefully curated educational videos, many of which represent collaborations betwixt talented educators and animators nominated through the TED-Ed website.
Feedback
Have feedback to give about this text? Click here.
Found a typo and want extra credit? Click hither.
Source: https://chem.libretexts.org/Courses/Oregon_Institute_of_Technology/OIT:_CHE_202_-_General_Chemistry_II/Unit_2:_Electrons_in_Atoms/2.2:_Atomic_Orbitals_and_Quantum_Numbers
0 Response to "What Information Is Needed to Determine the General Shape of an Orbital"
Postar um comentário